Materials Science: Unit I: Crystallography
Crystal Structures
The alkali metals Li, Na, K, etc., have bcc structures, some transition elements and rare earths have fcc structures, elements of second group have hcp structures.
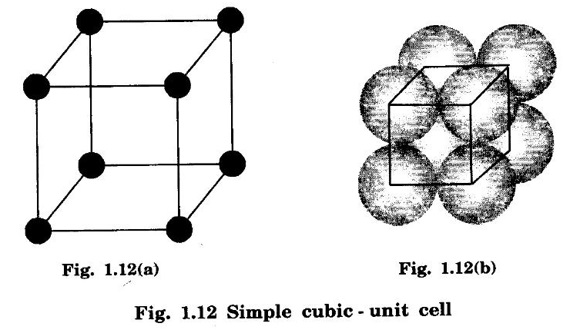
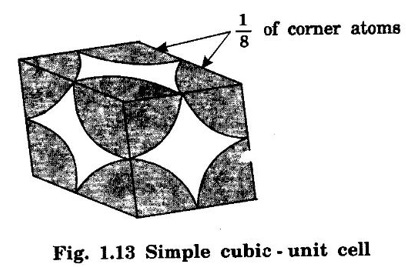
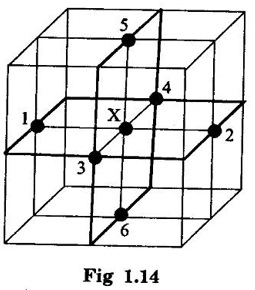
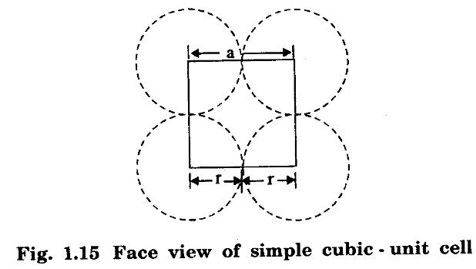
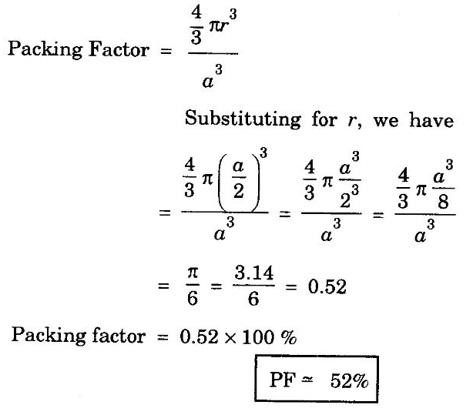
CRYSTAL STRUCTURES COORDINATION NUMBER AND PACKING FACTOR FOR CRYSTAL STRUCTURES SC, BCC, FCC and HCP It is noted that a large number of metallic crystal structures have hcp, fcc and bec structures. Simple Cubic (SC) structure is very rare in metals, The alkali metals Li, Na, K, etc., have bcc structures, some transition elements and rare earths have fcc structures, elements of second group have hcp structures. The simple cubic structure is the simplest and easiest crystal structure. In this structure, there is one atom at each of the '8' corners of unit cell. These atoms touch each other along cube edges. (fig 1.12). The unit cell of a simple cubic structure is shown in fig 1.12. The representative arrangement is drawn in fig. 1.12 (a). The actual way of arrangement is shown in Fig. 1.12 (b). There are 8 atoms, one atom at each corner of unit cell. Each corner atom is shared by 8 adjascent unit cells. (Fig. 1.13) ⸫ Share of each unit cell = 1/8 of corner atoms = 1/8 × 8 = 1 Total number of atoms in one unit cell = 1 atom Simple cubic unit cell has 8 corner atoms. Let us consider one of the corner atoms (say X). It is shared by 8 adjacent unit cells as shown in fig 1.14. There are 4 nearest neighbouring atoms to this particular atom X' which are shown by 1, 2, 3, and 4 in a plane (horizontal plane). Further, there are 2 more nearest atoms, one directly above (atom 5) and the other one directly below (atom 6) the atom X. Thus, there are only six (4+2) nearest neighbours for the atom X. Hence, coordination number for simple cubic is 6. Note: Similarly if we consider any corner atom, the total number of nearest neighbours i.e., the co-ordination number is the same. Consider a face of unit cell of a simple cubic structure (fig 1.15). The atoms touch each other along the edges of the cube. It is clear that the distance between the centres of two nearest atoms is equal to the cube edge a. If 'a' is the side of the unit cell and r its radius, then, from fig. 1.15, 2r = a Number of atoms per unit cell = 1 Volume of the atoms in the unit cell, v = 1 × 4/3 πr3 Atomic radius r = a/2 Total volume of the unit cell, V = a3 We know that packing factor = υ/V Substituting for v and V, we have Thus, 52% of the volume of unit cell is occupied by the atoms and the remaining 48% volume is vacant. Example: Only one element polonium (Po) at certain temperature range exhibits this crystal structure.Simple Cubic (SC) Structure
1. Number of atoms per unit cell
2. Coordination number
3. Atomic radius
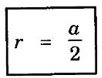
4. Packing factor
Materials Science: Unit I: Crystallography : Tag: : - Crystal Structures
Materials Science: Unit I: Crystallography
Under Subject
Materials Science
PH3251 2nd semester Mechanical Dept | 2021 Regulation | 2nd Semester Mechanical Dept 2021 Regulation
Related Subjects
Professional English II
HS3251 2nd Semester 2021 Regulation | 2nd Semester Common to all Dept 2021 Regulation
Statistics and Numerical Methods
MA3251 2nd Semester 2021 Regulation M2 Engineering Mathematics 2 | 2nd Semester Common to all Dept 2021 Regulation
Engineering Graphics
GE3251 eg 2nd semester | 2021 Regulation | 2nd Semester Common to all Dept 2021 Regulation
Physics for Electrical Engineering
PH3202 2nd Semester 2021 Regulation | 2nd Semester EEE Dept 2021 Regulation
Basic Civil and Mechanical Engineering
BE3255 2nd Semester 2021 Regulation | 2nd Semester EEE Dept 2021 Regulation
Electric Circuit Analysis
EE3251 2nd Semester 2021 Regulation | 2nd Semester EEE Dept 2021 Regulation
Physics for Electronics Engineering
PH3254 - Physics II - 2nd Semester - ECE Department - 2021 Regulation | 2nd Semester ECE Dept 2021 Regulation
Electrical and Instrumentation Engineering
BE3254 - 2nd Semester - ECE Dept - 2021 Regulation | 2nd Semester ECE Dept 2021 Regulation
Circuit Analysis
EC3251 - 2nd Semester - ECE Dept - 2021 Regulation | 2nd Semester ECE Dept 2021 Regulation
Materials Science
PH3251 2nd semester Mechanical Dept | 2021 Regulation | 2nd Semester Mechanical Dept 2021 Regulation
Basic Electrical and Electronics Engineering
BE3251 2nd semester Mechanical Dept | 2021 Regulation | 2nd Semester Mechanical Dept 2021 Regulation
Physics for Civil Engineering
PH3201 2021 Regulation | 2nd Semester Civil Dept 2021 Regulation
Basic Electrical, Electronics and Instrumentation Engineering
BE3252 2021 Regulation | 2nd Semester Civil Dept 2021 Regulation
Physics for Information Science
PH3256 2nd Semester CSE Dept | 2021 Regulation | 2nd Semester CSE Dept 2021 Regulation
Basic Electrical and Electronics Engineering
BE3251 2nd Semester CSE Dept 2021 | Regulation | 2nd Semester CSE Dept 2021 Regulation
Programming in C
CS3251 2nd Semester CSE Dept 2021 | Regulation | 2nd Semester CSE Dept 2021 Regulation















