Engineering Thermodynamics: Unit II: Second Law and Entropy
Solved Problem on Clausius Inequality
Second Law of Thermodynamics
Solved Problem on Clausius Inequality: Second Law and Entropy - Engineering Thermodynamics
SOLVED PROBLEM ON CLAUSIUS INEQUALITY Problem 2.11 An inventor claims that his new engine will develop 30 kW for a heat addition of 2400 kJ/min. The highest and the lowest temperature of the cycle are 2200°C and 327°C respectively. Would you agree his claim? Use Clausius inequality method. Given data: W = 30 kW = 30 × 60 = 1800 kJ/min Q1 = 2400 kJ/min T1 = 1527°C = 2200 + 273 = 2473 K T2 = 327°C = 327 + 273 = 600 K To find: Agreement his claim. Solution: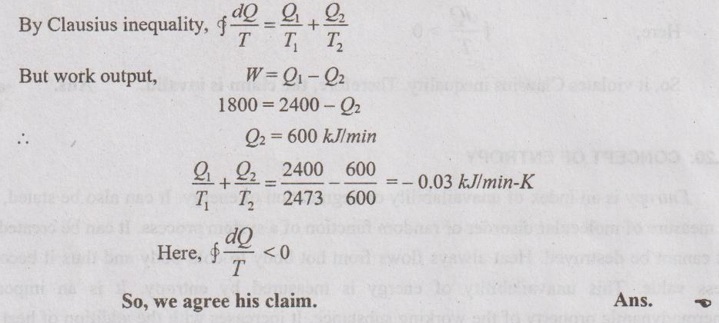
Engineering Thermodynamics: Unit II: Second Law and Entropy : Tag: : Second Law of Thermodynamics - Solved Problem on Clausius Inequality
Engineering Thermodynamics: Unit II: Second Law and Entropy
Under Subject
Engineering Thermodynamics
ME3391 3rd semester Mechanical Dept | 2021 Regulation | 3rd Semester Mechanical Dept 2021 Regulation
Related Subjects
Transforms and Partial Differential Equations
MA3351 3rd semester civil, Mechanical Dept | 2021 Regulation | 3rd Semester Mechanical Dept 2021 Regulation
Engineering Mechanics
ME3351 3rd semester civil, Mechanical Dept | 2021 Regulation | 3rd Semester Mechanical Dept 2021 Regulation
Engineering Thermodynamics
ME3391 3rd semester Mechanical Dept | 2021 Regulation | 3rd Semester Mechanical Dept 2021 Regulation
Fluid Mechanics and Machinery
CE3391 3rd semester Mechanical Dept | 2021 Regulation | 3rd Semester Mechanical Dept 2021 Regulation
Engineering Materials and Metallurgy
ME3392 3rd semester Mechanical Dept | 2021 Regulation | 3rd Semester Mechanical Dept 2021 Regulation
Manufacturing Processes
ME3393 3rd semester Mechanical Dept | 2021 Regulation | 3rd Semester Mechanical Dept 2021 Regulation





